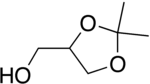
Solketal
| |
| Names | |
|---|---|
| Preferred IUPAC name
(2,2-Dimethyl-1,3-dioxolan-4-yl)methanol | |
| Other names
Isopropylidene glycerol
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.002.626 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H12O3 | |
| Molar mass | 132.159 g·mol−1 |
| Appearance | clear colorless liquid |
| Density | 1.063 g/mL at 25 °C |
| Boiling point | 188 to 189 °C (370 to 372 °F; 461 to 462 K) |
| Miscible | |
| Solubility | Miscible in most organic solvents (alcohols, ethers, hydrocarbons) |
| Hazards | |
| Flash point | 80 °C (176 °F; 353 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Solketal is a protected form of glycerol with an isopropylidene acetal group joining two neighboring hydroxyl groups. Solketal contains a chiral center on the center carbon of the glycerol backbone, and so can be purchased as either the racemate or as one of the two enantiomers. Solketal has been used extensively in the synthesis of mono-, di- and triglycerides by ester bond formation. The free hydroxyl group of solketal can be esterified with a carboxylic acid to form the protected monoglyceride. The isopropylene group can then be removed using an acid catalyst in aqueous or alcoholic medium. The unprotected diol can then be esterified further to form either the di- or triglyceride.
Another route to specific di- or triglycerides involves converting the solketal to glycidol (2,3-epoxy-1-propanol) and esterifying this with one fatty acid before opening the epoxy by heating in the presence of a second fatty acid and a catalyst. This second fatty acid is put on the third carbon atom, and then a third fatty acid can be added to the second carbon atom.[1]
References[edit]
- ^ C.M. Lok, J.P. Ward, D.A. van Dorp (Mar 1976). "The synthesis of Chiral Glycerides starting from D- and L-serine". Chemistry and Physics of Lipids. 16 (2): 115–122. doi:10.1016/0009-3084(76)90003-7. PMID 1269065.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
This article includes a list of references, related reading, or external links, but its sources remain unclear because it lacks inline citations. (July 2013) |
- Mary Renoll and Melvin S. Newman (1955). "dl-ISOPROPYLIDENEGLYCEROL". Organic Syntheses. 28: 73; Collected Volumes, vol. 3, p. 502.
- Sanderson, John R.; Lin, Jiang J.; Duranleau, Roger G.; Yeakey, Ernest L.; Marquis, Edward T. (1988). "Free radicals in organic synthesis. A novel synthesis of glycerol based on ethylene glycol and formaldehyde". Journal of Organic Chemistry. 53 (12): 2859. doi:10.1021/jo00247a043.
- "Solketal". Logo of chemBlink Inc. Online Database of Chemicals from Around the World. Archived from the original on 13 December 2010.
- Matsumoto, Yoshihiko; Mita, Keisuke; Hashimoto, Keiji; Iio, Hideo; Tokoroyama, Takashi (1996). "Selective cleavage of ethers using silica-alumina gel catalysts prepared by the sol-gel method". Tetrahedron. 52 (28): 9387. doi:10.1016/0040-4020(96)00501-7.
